ACPGBI supports its members through sponsorship and curation of a number of high quality audits and databases.
Ileal Pouch Registry
The Ileoanal Pouch Registry was originally established by Professor John Nicholls and gathered information on over 5300 patients who have undergone pouch surgery. Maintenance costs associated with the secure platform, website access, clinical governance and curation of a pouch report were kindly sponsored by IA, the Ileostomy and Internal Pouch support group.
In 2018 it became clear that the pouch registry needed to be upgraded and the website modernised. This was for various reasons including
- The need for enhanced governance with the advent of GDPR
- Desire of a patient reported outcome element
- Integration with the IBD registry run by gastroenterologists

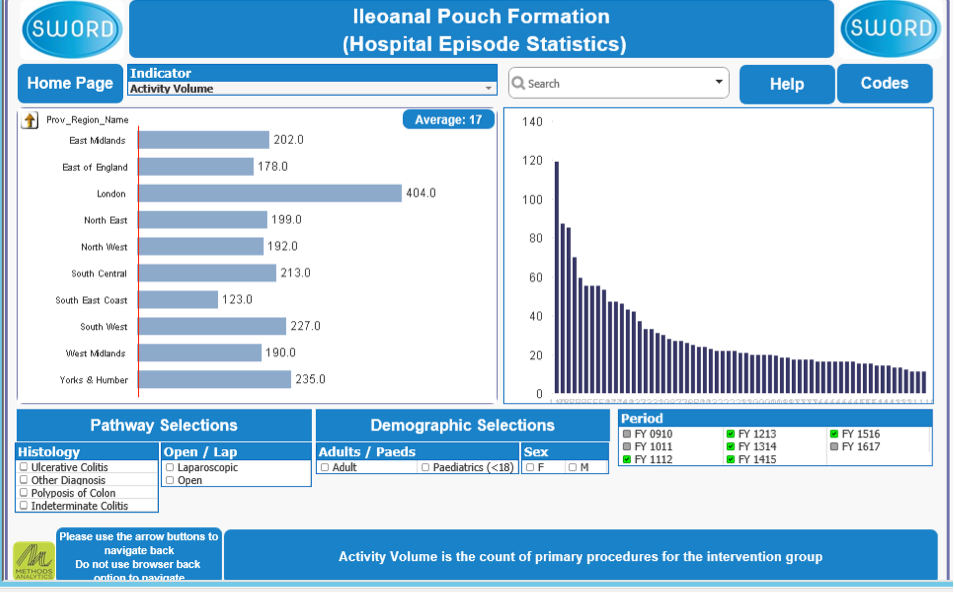
Surgical Workload Outcomes Audit Database (SWORD)
The ACPGBI has commissioned, funded and collaborated on a pouch surgery module on SWORD for its members. SWORD relies on HES data and so this will only be available for England in the first instance.
The database demonstrates activity, indication, proportion laparoscopic, gender, year, length of stay, readmission and reoperation rates. It has the utility of drilling down to regional, Trust and individual level. SWORD will be available to ACPGBI Consultant members who will be given their own pseudo-anonymised code (based on GMC number) so that individual surgeons are protected from identification by other viewers.
The IBD CAG has also been exploring development of further IBD surgery reporting tools on the SWORD platform and the following IBD modules are currently in preparation, in addition to pouch surgery:
- Colectomy
- Proctectomy and proctocolectomy with permanent ileostomy
- Crohn’s small bowel resection and strictureplasty
- Crohn’s ileocaecal resection
- Perianal Crohn’s procedures
IBD Clinical Advisory Group
The Inflammatory Bowel Disease (IBD) Clinical Advisory Group (CAG) provides oversight of initiatives and finances for IBD projects within ACPGBI. The CAG has been responsible for providing guidance on the Pouch Registry and SWORD, but is also now working on fellowships, accreditation and guidelines in IBD Surgery. The IBD CAG is chaired by Professor Steven Brown and represents multidisciplinary interests, including BSG, IA and CCUK.
Anal Cancer Database
The National Anal Cancer Database has been commissioned by ACPGBI and built by Dendrite Clinical Systems. The Clinical Leads are Professor Andrew Renehan (Chair) and Miss Karen Nugent (Deputy).
The database is intended primarily as an audit tool to allow anal cancer MDTs to monitor their activity and performance against other units, thereby driving standards in the management of patients with anal cancer. It is not intended as a primary research tool, although it may act as a research information resource for trial recruitment of specific patient groups.
A pilot has been run on the Dendrite platform. As the database includes patient-reported outcome measures, entered by patients directly, ethical approval is required and so an MRA application will be needed to support this aspect of the database prior to its widespread implementation.
Anal Cancer Database website
International Colorectal Stent Registry
The aim of this registry is to collect individual patient records and track these procedures as a bridge to surgery and for palliative care.
International Colorectal Stent Registry website
National Audit of Small Bowel Obstruction
 The National Audit of Small Bowel Obstruction (NASBO) will run prospectively from 13 January 2017 after a successful pilot in late 2016. NASBO is a joint collaborative-led venture between ACPGBI and the South Yorkshire Surgical Research Group and West Midlands Research Collaborative. Matthew Lee and Adele Sayers of SYSuRG are the collaborative leads.
The National Audit of Small Bowel Obstruction (NASBO) will run prospectively from 13 January 2017 after a successful pilot in late 2016. NASBO is a joint collaborative-led venture between ACPGBI and the South Yorkshire Surgical Research Group and West Midlands Research Collaborative. Matthew Lee and Adele Sayers of SYSuRG are the collaborative leads.
Our main reasons for promoting NASBO are to support the excellent work carried out by the research collaboratives and to build a network of research collaborators in all acute trusts for the Delphi research programme.
Bowel Research UK is the principal sponsor of NASBO with additional pledges of financial support and steering group commitment from the Royal College of Surgeons of England, the Royal College of Surgeons of Edinburgh, and the Royal College of Anaesthetists, as well as stakeholder professional societies including Association of Upper Gastrointestinal Surgeons, BASO – The Association for Cancer Surgery, British Association for Parenteral and Enteral Nutrition, British Society of Gastroenterology, Getting It Right First Time and National Emergency Laparotomy Audit.
The Sheffield Clinical Trials Research Unit is supporting NASBO. Over 150 hospitals and Trusts have signed up to participate in NASBO. Anticipated data capture is between 1000 and 5000 patients over the two month data collection period.
NASBO Steering Group member list for 2016
NASBO website
National Complicated Acute DiverticulitiS (CADS) Audit
ACPGBI supports the National Complicated Acute DiverticulitiS (CADS) Audit looking at management of patients admitted with acute diverticulitis. The protocol was published in BMJ Open Gastro 2016 3 e000117. The audit aims to collect data on the national incidence, management strategies, short-term (3 months) and long-term (2 years) outcomes, stoma rates, and the role of surgical and radiological interventions. The initial recruitment phase took place over three months in 2014 and final follow-up was due in late 2016.
CADS Audit protocol
National Lower Gastrointestinal Bleed Audit
ACPGBI supported the National Lower Gastrointestinal Bleed (NLGB) Audit which was jointly funded by Bowel Research UK and NHS Blood and Transplant.
The protocol was published in BMJ Open 2016 6 e011752 and the results of the audit have now been published online.
NLGB Audit protocol
NLGB Audit results 2016
National Emergency Laparotomy Audit
The National Emergency Laparotomy Audit (NELA) is a well-established prospective audit run by the Royal College of Anaesthetists in conjunction with surgical and other key stakeholders. The aim of NELA is to provide high quality comparative data from all providers of emergency laparotomy to facilitate improvements in care for patients. The National Surgical Lead for NELA is Mr Iain Anderson.
The ACPGBI is currently collaborating with NELA on analysis of patients who have undergone colectomy (ACPGBI Lead Mr Richard Guy) and Hartmann’s procedure (ACPGBI Lead Mr Hugh Paterson).
NELA Second Patient Report
UK Colorectal Cancer Repository (CORECT-R)
Professor Eva Morris’ application to CRUK for the UK COloRECTal cancer Repository (CORECT-R), a shared repository of all colorectal cancer databases, was supported by ACPGBI and NBOCA, and has been successful with an award of £3.4 million.